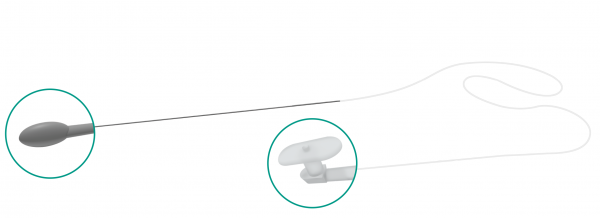
Masterka®
Description :
Masterka® is one of FCI latest innovation for treatment of congenital nasolacrimal duct obstruction resistant to probing.
Unlike the traditional “pulled” technique in which the stent is advanced through the nasolacrimal system and retrieved through the nose by pulling on the guide probe or thread, the Masterka® has no metallic probe or suture attached to it and, therefore, it is not pulled out of the nose.
Instead, the Masterka® is pushed into the nasolacrimal duct and anchored in place at the punctum by a plug-like fixation head.
Main characteristics :
• Easy to insert
• No nasal retrieval
• No knots
• No sutures
• Less traumatic
• Requires sizer (S1.1289) to select the length & 0.3mm disposable punctumdilator and plug inserter (S1.3090)
• Available in 3 lengths: 30, 35 and 40mm
• 3mmplug collarette
• Medical grade silicone
• Sterile